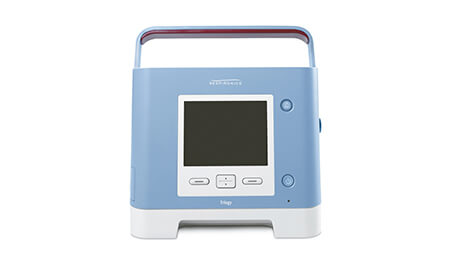
Medical Device Recall Information
Philips Respironics Sleep and Respiratory Care devices
In June 2021, after discovering a potential health risk related to a part in certain CPAP, BiPAP and Mechanical Ventilator devices, Philips issued a voluntary Field Safety Notice (outside U.S.) / voluntary recall notification (U.S. only).
Patient safety is our top priority, and we are committed to supporting our patients, durable medical equipment providers (DMEs), distributors, home health partners, and clinicians through the complete remediation process.
Throughout the remediation of this recall we will provide guidance and share next steps so you can ensure you have the most current and accurate information. We thank you for your patience as we work to restore your trust.
Not registered?
Additional information for…

Patient


Questions and answers
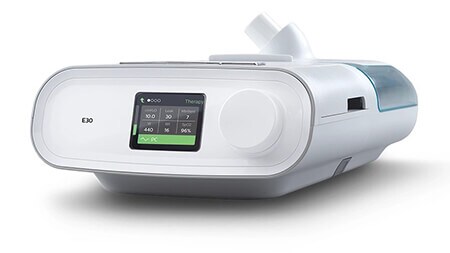
E30
(Emergency Use Authorization)

DreamStation ASV
Also known as DreamStation BiPAP autoSV

DreamStation ST, AVAPS
Also known as DreamStation BiPAP AVAPS, DreamStation BiPAP S/T

SystemOne ASV4
Also known as System One BiPAP autoSV, System One BiPAP autoSV Advanced

C Series S/T, AVAPS
Also known as System One BiPAP AVAPS (C-Series), System One BiPAP S/T (C-Series)

OmniLab Advanced Plus
In-Lab Titration Device
Continuous Ventilator, Minimum Ventilatory Support, Facility Use

A-Series BiPAP Hybrid A30
Also known as BiPAP Hybrid A30Ventilator (A-Series)
(not marketed in US)

A-Series BiPAP V30 Auto Ventilator
Also known as BiPAP V30 Auto Ventilator (A-Series)
Continuous Ventilator, Non-life Supporting

A-Series BiPAP A40
Also known as BiPAP A40 Ventilator (A-Series)
(not marketed in US)

A-Series BiPAP A30
Also known as BiPAP A30 Ventilator (A-Series)
(not marketed in US)
If your device is affected...
What products are not affected and why?
Products that are not affected may have different sound abatement foam materials, as new materials and technologies are available over time. Also, sound abatement foam in unaffected devices may be placed in a different location due to device design.